Research to help Operators determine their Noro-E.coli-hep A risk factor. This definition of a Clean Hand is an articulation in search of the science: A Clean Hand in the foodservice and food processing industries is one that is unlikely to transfer pathogens from the hand to food, surfaces or directly to people. Handwashing For Life® […]
Food Safety Research Reveals New Handwashing Heroes
Common ground drives budget sharing to defeat Norovirus Too many times needed food safety research is buried by either a smothering budget or by an agenda to promote an individual position. In April of 2023, Handwashing For Life® joined Ohio State’s Center for Foodborne Illness Research & Prevention in submitting issue I-016 to the FDA’s […]
Comparing Pathogen Transfer Rates: Handwash vs. Hand Sanitizer
Well before COVID, the FDA raised the suggested handwash time from “10-15” to 20 seconds. During COVID, the CDC reinforced the 20-second variant but without the science to support the change. Now that the COVID alarm stage is over, can restaurants go back to 15 seconds? There is also a lack of clarity on the […]
The Operator’s Choice Handwash
A new evidence-based standard for a Clean Hand Many well-intentioned advisers have failed to help operators change handwashing behaviors in Foodservice. Their vague advice, starting with the FDA’s Model Food Code, is not actionable. A synonym for vague is lacking definition and surprisingly, there is no definition for a Clean Hand. These 7 steps are […]
Hand Sanitizer for Visually Soiled Hands: SaniTwice® & SaniOnce™
Alcohol has an extensive history in safely reducing the risk of infections. There are references back to biblical times of using wine to cleanse wounds. Its success as a hand sanitizer was confirmed during the global COVID experience, and recommended for use where hands are not visually contaminated. Restaurant clientele now expect to have ready […]
Clean Hand Research: Development Status
Research Benefits: Handwashing For Life® is taking a deep dive into hand hygiene research. Its expected deliverable is an enabling of operators to make decisions based on evidence rather than the commonly compromised and inconsistent guidelines from well-meaning propagators of myths, the status quo, and the science of past centuries. A Clean Hand in the […]
Hand Washing For Life®: Research Pivot
Handwashing For Life®, for its 25-year history, has been funded by its Leadership Forum Members, working together in the interests of public safety. We are forever grateful to them for their mission-critical contribution in establishing Handwashing For Life’s body of knowledge. This resource of Best Practices lives on to help operators seek, find, and understand […]
Handwashing For Life® Adds Research Capabilities
Handwashing For Life’s (HFL) over-arching goal of “Overcoming Underwashing” was advanced by HFL’s merger with Ohio State’s Center for Foodborne Illness and Prevention (CFI). Handwashing For Life’s founder and lead scientist, Jim Mann, continues his pursuit of evidence-based data for operators to make more informed policy and investment decisions. Research projects will start small by […]
International Clean Hands Week
September 19-25, 2022 Please join this annual campaign, sponsored by the CDC-facilitated Clean Hands Coalition. Raise awareness of the dangers of poor hand hygiene. Check out a variety of ideas to improve the away-from-home wellness factor in everyone’s life. Clean hands are the cure to the spread of colds, the flu, and those illnesses acquired […]
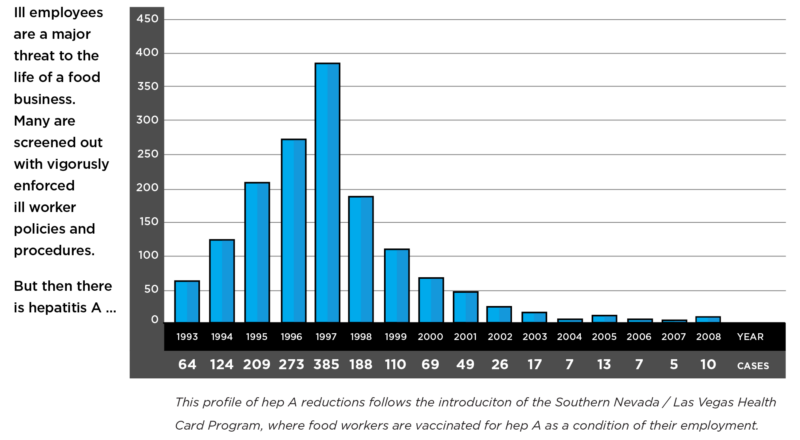
The 1-2 Punch For Brand Protection From Hep A Outbreaks
Brand Damage Grossly Exceeds Out-of-pocket Immune Globulin Treatment Costs The first punch … Handwashing. Missed hand washes are frequently at the heart of Hep A outbreaks in Foodservice and Healthcare settings, especially in restaurants and nursing homes. Infected food handlers, including caregivers, commonly transfer this highly contagious pathogen.Fecal-hand-oral is the primary transmission path. Most infections result from direct contact or […]